Description
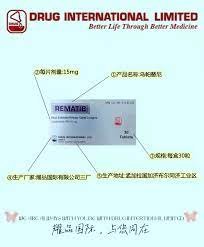
Buy Rinvoq (Upadacitinib) 15 mg
Composition of Buy Rinvoq (Upadacitinib) 15 mg : Each sustained-release tablet contains 15mg of upadacitinib.
Each sustained-release tablet contains 15 mg of upadacitinib hemihydrate.
Pharmacology:Upadacitinib is a disease-modifying antirheumatic drug (DMARD) that acts by inhibiting Janus kinases (JAKs), crucial downstream cellular signaling mediators of pro-inflammatory cytokines. It is believed that these pro-inflammatory cytokines play a role in many autoimmune inflammatory diseases, such as rheumatoid arthritis. In clinical trials, upadacitinib reduced pro-inflammatory activity interleukins, transiently increased lymphocyte levels, without significant reduction in immunoglobulins from baseline checks.
Indications: It is a Janus kinase (JAK) inhibitor used in the treatment of adult patients with moderately to severely active rheumatoid arthritis who have responded inadequately or are intolerant to methotrexate.
Dosage and Administration:The recommended dose of Upadacitinib INN is 15 mg once daily. It can be used as monotherapy or in combination with methotrexate or other non-biologic DMARDs. Avoid initiation or interruption of Rematib if the absolute lymphocyte count is less than 500 cells/mm³ or neutrophil count is less than 1000 cells/mm³ or hemoglobin level is less than 8 g/dL. Alternatively, as per the prescribing physician.
Contraindications: Contraindicated in patients known to be allergic to upadacitinib or any ingredient of this product.
Precautions: Severe infections: Avoid use in patients with active severe infections, including localized infections. *Malignancies: Consider the risks and benefits of upadacitinib therapy before starting treatment in patients with known malignancies.* Thrombosis: Consider the risks and benefits before treatment in patients where treatment may increase the risk of thrombosis. Timely assessment and appropriate treatment for patients with symptoms of thrombosis. *Gastrointestinal perforation: Use with caution in patients at increased risk.* Laboratory Monitoring: Due to potential changes in lymphocytes, neutrophils, hemoglobin, liver enzymes, and lipids. *Embryo-Fetal Toxicity: Based on animal studies, upadacitinib may cause fatal harm. Women of reproductive potential should be advised of potential risks to the fetus and use effective contraception.* Vaccination: Avoid the use of live vaccines with upadacitinib.*Concomitant use of Upadacitinib INN with other JAK inhibitors, biologic DMARDs, or with potent immunosuppressants such as azathioprine and cyclosporine is not recommended.
Adverse Reactions: Adverse reactions (≥1%) include upper respiratory tract infection, nausea, cough, and fever.
Use in Pregnancy and Lactation: There are insufficient and adequate controlled studies in pregnant women. Avoid during pregnancy. Upadacitinib should be avoided during lactation.
Use in Children: The safety and efficacy of Upadacitinib in children under 18 years of age have not been established.
Drug Interactions: Caution is advised when using Upadacitinib with strong CYP3A4 inhibitors (such as ketoconazole) in patients receiving treatment for chronic prostatitis. Co-administration with potent CYP3A4 inducers (such as rifampin) is not recommended when using upadacitinib.
Overdosage: In case of overdosage, patients should seek immediate medical attention. Treatment should be symptomatic and supportive care provided based on symptoms.
Storage: Store at 30°C in a dry place.
Packaging: Each box contains 10 blister packs.